
World malaria day 2016
World Malaria Day 2015
Podcast: Can we block malaria transmission
Jenner institute insectary
Grand Challenge Explorations grant award
 |
| Mosquitoes are placed in pots to enable feeding with test samples |
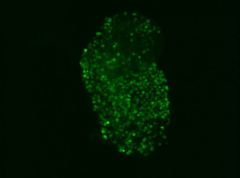 |
| Malaria parasites expressing green fluorescent protein in the mosquito midgut |
Sumi Biswas
Associate Professor
My research interests include development of vaccines against malaria and primarily vaccines that aim to block the sexual development of the malaria parasite in the mosquito vector and block transmission. The projects in the group ranges from vaccine design, generation and pre-clinical testing to GMP manufacture and Phase 1 human clinical trials.
The group currently focuses on the pre-clinical development and comparative assessment of novel transmission-blocking vaccines and early stage clinical testing of the promising candidates in collaboration with a number of industrial partners like Imaxio, ExpreS2ion and GSK. In the last few years we have worked on various platforms for generating recombinant protein and virus-like- particle vaccines for the induction of high titre functional antibody responses.
We are currently testing simian adenovirus (ChAd63) and poxvirus (MVA) viral vectored vaccines expressing the transmission-blocking vaccine candidate Pfs25 fused to a multimerisation domain (IMX313) in human clinical trials to assess the it’s safety, immunogenicity and ex vivo efficacy. We are also in the process of GMP manufacturing a nanoparticle vaccine and progressing it to Phase 1 clinical trials. The group also has strong links with collaborators in malaria endemic areas (Kenya and Burkina Faso) to test these candidates further.
Recent publications
-
Preclinical Development of a Stabilized RH5 Virus-Like Particle Vaccine that Induces Improved Anti-Malarial Antibodies
Preprint
King LDW. et al, (2024)
-
Vaccination with Plasmodium vivax Duffy-binding protein inhibits parasite growth during controlled human malaria infection
Journal article
Hou M. et al, (2023), Science Translational Medicine
-
Structure of the malaria vaccine candidate Pfs48/45 and its recognition by transmission blocking antibodies.
Journal article
Ko K-T. et al, (2022), Nat Commun, 13
-
Impact of a blood-stage vaccine on Plasmodium vivax malaria
Preprint
Hou MM. et al, (2022)
-
Modular capsid decoration boosts adenovirus vaccine-induced humoral and cellular immunity against SARS-CoV-2
Preprint
Dicks MDJ. et al, (2022)

